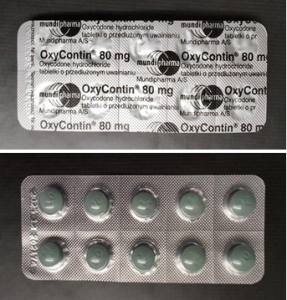
The National Agency for Food and Drug Administration and Control (NAFDAC) has issued a warning about a batch of falsified OXYCONTIN 80 mg (oxycodone hydrochloride) tablets found in Switzerland’s unregulated market.
The genuine manufacturer, Mundipharma, reported it to the World Health Organisation (WHO) in February 2025, saying that the fake product imitates OxyContin 80 mg authorised for sale in Poland.
Laboratory analysis by the Drug Information Centre in Zurich revealed that the falsified tablets contained a potent synthetic opioid suspected to be a nitazene compound rather than oxycodone.
Nitazenes, including derivatives like metonitazene and isotonitazene, are far more potent than oxycodone and carry a high risk of overdose and death, especially when combined with other depressants.
Mundipharma confirmed the product was falsified and not manufactured by them. The WHO warned the fake tablets misrepresent their identity, composition, and source, posing serious health risks, particularly to individuals with substance use disorders.
Visual differences include incorrect placement of the batch number and expiry date on the blister strip. The counterfeit product lists batch number 262174 and expiry date December 2025.
NAFDAC has directed its zonal directors and state coordinators to intensify surveillance and remove any falsified products found in Nigeria. The agency urges importers, healthcare professionals, and consumers to verify medical products and buy only from licensed suppliers.
Suspicious products or side effects should be reported to NAFDAC offices via phone at 0800-162-3322, email at sf.alert@nafdac.gov.ng, or through the Med-safety app and NAFDAC’s website.
